Lungworms
Back to Fact Sheets
What is lungworm disease?
Lungworms can affect both dogs and cats. Angiostrongylus vasorum and Crenosoma vulpis affect mainly dogs, while Aelurostrongylus abstrusus is found in the cat. These parasites commonly localise in the cardiopulmonary system. Currently there is strong evidence for A. vasorum spread in the UK, therefore we will focus on this specific lungworm.
Download PDF
How is the disease transmitted?
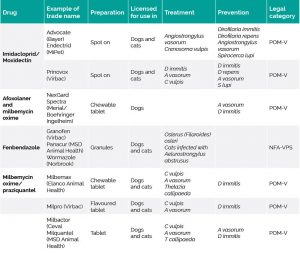
Dogs are infected by ingesting infected slugs and snails containing mature L3 larvae. The larvae develop in the slugs and snails from L1 to L3.
Dogs eat the snails and/or slugs, then the L3 larvae enters the portal circulation through the intestinal wall and migrates to the abdominal lymph nodes and moults to L4. They migrate through the liver parenchyma and, eventually, reach the right ventricle and pulmonary arteries, where they finish their development as adults – this is their final location.
The adult parasites are located in the right side of the heart and the pulmonary arterial circulation, smaller parasites are often found in the small arteries in the periphery of the lungs. The larvae that hatches from an egg crosses into the alveoli and are carried to the pharynx and swallowed, to pass out in the faeces.
What is the Pathogenesis of the disease?
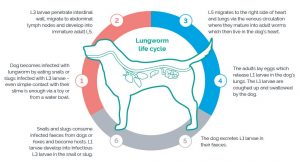
The presence of adult nematodes in the pulmonary vasculature leads to an immunological response that is characterized by immune complex deposition. This immune complex deposition may also result in uveitis, chorioretinitis and glomerulonephritis
Damage to the vascular endothelium leads to activation of the coagulation and inflammatory cascades, which results in a thrombosing arteritis within the affected vessels which can potentially result in disseminated intravascular coagulation. Acute intracranial haemorrhage could also happen as a result of a bleeding diathesis.
Simultaneous recruitment of inflammatory cells in the surrounding pulmonary interstitium results in interstitial pneumonitis. Pulmonary inflammation is exacerbated by the emergence of L1 larvae from the pulmonary capillary bed into the alveolar air spaces. The severity of clinical signs depends on the dose of parasites.
L1 larvae have also been retrieved from the left chambers of the heart and may embolise to remote capillary beds. Acute-onset lumbar pain and paraparesis have been reported and are thought to occur secondarily to embolisation of larvae.
Finally, chronic infection results in the more typical clinical signs of coughing, progressive dyspnea and exercise intolerance as a consequence of granulomatous inflammation and fibrosis of the lungs.
What are the most common clinical signs?
The most common clinical presentation is cardiorespiratory, such as coughing – either alone or in combination with bleeding disorders or neurological signs.
The most common signs would be coughing, dyspnea, exercise intolerance, epistaxis, petechiae, ecchymoses, haemoptysis to syncope, bleeding to any body cavity and pulmonary hypertension among others.
The most common neurological signs are variable depending on the location of the bleeding. These could include from seizures or cerebellar signs, to spinal signs (paresis or plegia).
The respiratory, coagulation and neurological problems might not all happen in the same patient or at the same time.
How can we diagnose lungworms?
Blood tests may be indicative of a lungworm infestation; commonly associated with an eosinophilia and hyperglobulinemia. Prolonged coagulation times and a reduction in platelet numbers is usually attributable to a chronic disseminated intravascular coagulation (DIC). Regenerative anaemia and hypercalcemia have also been reported with lungworm infection.
There is a specific test “Angiodetect” which detects the circulating parasite antigen in blood, is easy to use and the results are obtained in less than 15 minutes. A positive result is very reliable, but a negative result in a case highly suspicious for disease should be followed up with an alternative diagnostic test or retested at a later date.
A definitive diagnosis of angiostrongylosis can be obtained by demonstrating the presence of L1 larvae in faeces using the Baermann sedimentation technique. A.vasorum L1 larvae should be differentiated from other canine lungworm larvae based on their morphological characteristics.
Imaging is a very useful diagnostic tool. Pulmonary haemorrhage can be observed by an MRI of the spine or the brain.
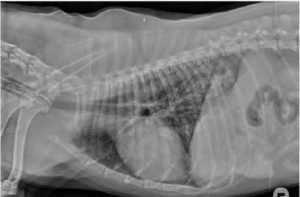
Lateral chest view with diffuse and severe interstitial pattern in a dog positive for lungworm
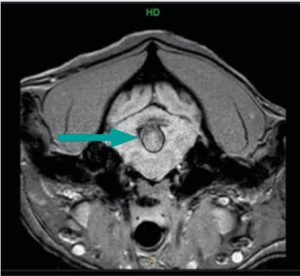
Transverse image over the cerebellum GRE. See the intraxial bleeding
How can we treat and prevent lungworms?
There are two products currently licensed to treat A. vasorum infection: imidacloprid/moxidectin in combination and milbemycin oxime. However, fenbendazole is also widely used, despite the lack of a licensed indication for the treatment of A. vasorum. The reason for this is unknown but it may be due to a lower perceived risk of anaphylaxis from rapid worm kill.
Imidacloprid/ moxidectin and milbemycin oxime are both licensed preventive drugs when applied on a monthly basis. It is also recommended to keep dogs up to date with a full worming regimen, dispose of faeces daily, consider reducing the access of foxes to gardens and reduce contact with slugs.